Informing on health and wellness news in Laos
Provided by AGPLatest Findings from DME AWARE Delphi Study Presented at ARVO 2026 Annual Meeting
- Key findings from the DME AWARE initiative highlight the importance of non-invasive treatment options for early intervention in diabetic macular edema (DME) management
- Oculis is on track to report topline results in June 2026 from DIAMOND Phase 3 registrational trials with OCS-01, which aims to be the first eye drop therapy for DME
ZUG, Switzerland, May 05, 2026 (GLOBE NEWSWIRE) -- Oculis Holding AG (Nasdaq: OCS / XICE: OCS) (Oculis), a global biopharmaceutical company focused on breakthrough innovations to address significant unmet medical needs in ophthalmology and neuro-ophthalmology, today announces that the latest findings of the DME AWARE Delphi study were presented at the Association for Research in Vision and Ophthalmology (ARVO) 2026 Annual Meeting.
DME affects mainly a working-age population and is typically diagnosed at an average age of 52 years.1,2 One year following diagnosis, 60% of patients are not treated2a while we know that delaying treatment is not beneficial for patients as it represents a missed opportunity to fully regain vision. Furthermore, patients with less severe and early-stage DME are not often treated due to the risk/benefit ratio of invasive therapy options.3 Of those patients that are treated with an anti-VEGF, 40% of them have an inadequate treatment response defined as BCVA gain < 5-letters.4
DME AWARE, sponsored by Oculis, is a global Delphi initiative composed of a steering committee and panel of 25 leading retina and ophthalmology experts. The study aims to better understand the current DME landscape and to improve DME patient care by establishing a global expert consensus on unmet needs in DME management. The panel of experts answered three iterative anonymous surveys that posed critical questions about unmet needs in DME patient management, disease detection and assessment, and considerations for intervention.
Key findings from the DME AWARE Delphi initiative corroborated prior knowledge and suggest that unmet priority needs in DME include non-invasive treatment options, therapeutic options for early intervention, and novel therapies for concomitant use with current standard of care treatments. The goals of early intervention have been identified as improving or stabilizing visual function and reducing inflammatory factors. Furthermore, experts defined “early detection” as the detection of patients with DME prior to the presence of fluid and “early intervention” as the treatment of patients with DME prior to functional deterioration.
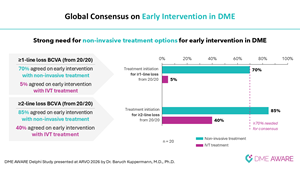
New findings from the third and final survey reinforce the strong need for non-invasive treatment options for early intervention in DME and showed consensus to treat with an eye drop in early intervention.
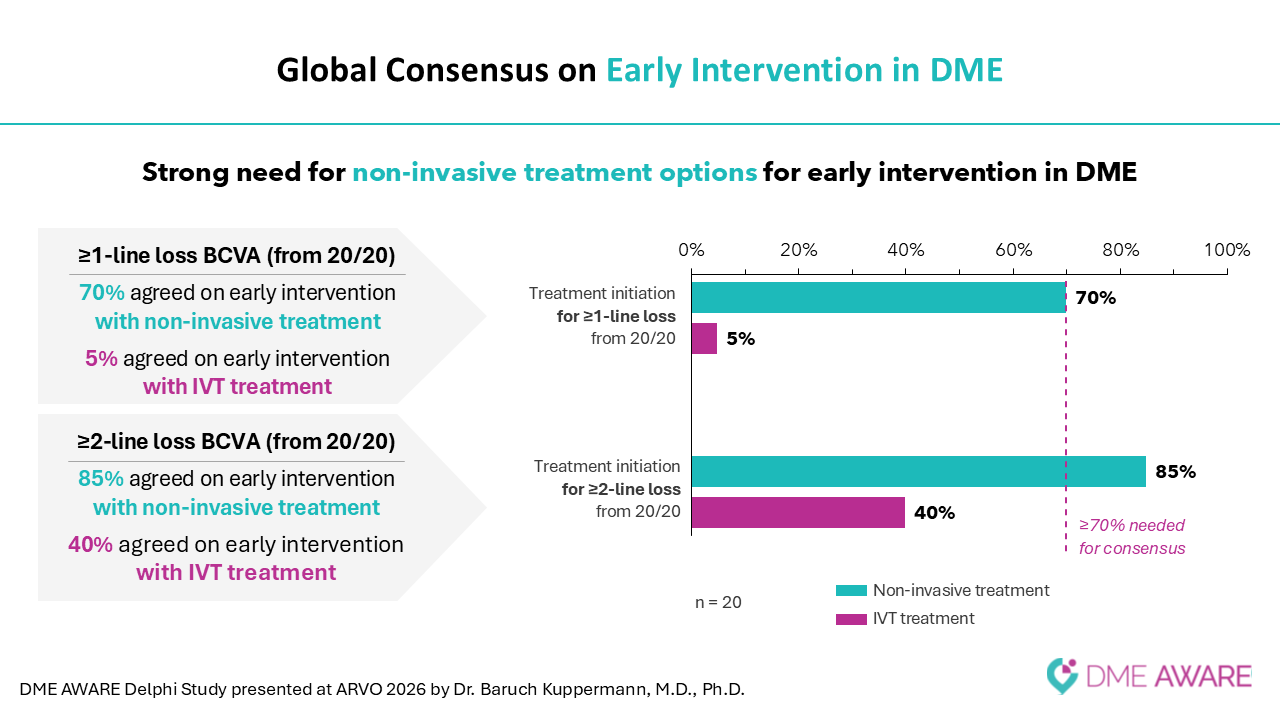
These findings underscore the critical unmet needs in DME patient management including non-invasive therapies for early intervention and concomitant use with current standard of care.
Professor Baruch D. Kuppermann, M.D., Ph.D., Department of Ophthalmology and Visual Sciences, University of California, Irvine and Gavin Herbert Eye Institute, said: “It was an honor for me to present these overall findings of the DME AWARE study, which reflect a truly global expert consensus on some of the most pressing challenges in DME management. The results underscore the ongoing interplay between the clinical need for effective disease control to prevent vision loss and the challenges associated with current treatment approaches. They also highlight growing interest among leading retinal and ophthalmology specialists in how less invasive options, if proven effective, could help address significant unmet needs remaining across different patient populations and stages of disease. The Delphi initiative lays important groundwork for how we can shape the future clinical practice to meaningfully improve patient care and treatment outcomes.”
Riad Sherif, M.D., Chief Executive Officer of Oculis, said: "While DME typically impacts patients at age 52, 60% remain untreated after one year due to the invasive nature of current therapies. As we approach the June readout of the pivotal DIAMOND trials, our Delphi survey confirms that a topical therapy could transform this landscape. OCS-01, a potential first-in-class eye drop, offers a non-invasive path for early intervention and a critical alternative for the 40% of patients who respond inadequately to injections."
- ENDS -
About OCS-01 eye drops and the OPTIREACH® technology
Leveraging Oculis’ proprietary technology, OCS-01 is an OPTIREACH® formulation of high concentration dexamethasone eye drop. It is being developed as an eye drop to treat the retina to offer a non-invasive treatment for diabetic macular edema (DME). This route of administration enables easy access to treatment in the early stages of the disease and can be used in combination with other therapies in later stages. In contrast, all currently available treatments require invasive delivery methods, such as intravitreal injections or ocular implants, to reach the retina. The OPTIREACH® solubilizing formulation technology addresses the main limitations of conventional eye drops by improving the solubility of lipophilic drugs, increasing the residence time on the eye surface and thereby, enabling the drug passage from the eye surface to the posterior segment of the eye. Oculis’ OCS-01 is being developed with the aim to transform the current treatment paradigm in DME as a non-invasive topical treatment option.
OCS-01 is an investigational drug in Phase 3 that has not received regulatory approval for commercial use in any country.
About Diabetic Macular Edema (DME)
DME, a complication from diabetes, is a leading cause of visual loss and legal blindness in working-aged adults. Currently, it is estimated to affect around 37 million people worldwide and, with the rise of diabetes, the prevalence is expected to increase to 53 million by 20405,6. DME is an irreversible and progressive complication of diabetic retinopathy and is related to consistently having high blood sugar levels that damage nerves and blood vessels in the macula, the area of the retina responsible for sharp vision. DME occurs when blood vessels in the retina swell, and then leak, leading to a fluid build-up (edema) into the retina. Despite available therapies, significant unmet medical needs remain for earlier treatment intervention or for inadequate responders to the current standard of care.
About Oculis
Oculis is a global biopharmaceutical company (Nasdaq: OCS; XICE: OCS) focused on breakthrough innovations to address significant unmet medical needs in neuro-ophthalmology and ophthalmology. Oculis’ highly differentiated late-stage clinical pipeline includes three core product candidates: OCS-01, an eye drop in pivotal registration studies, aiming to become the first non-invasive topical treatment for diabetic macular edema (DME); Licaminlimab, a novel, topical anti-TNFα in registrational trial, which is being developed with a genotype-based approach to drive precision medicine in dry eye disease (DED), and Privosegtor, a breakthrough neuroprotective candidate in the PIONEER program which consists of studies intended to support registration plans for treatment in optic neuropathies like optic neuritis (ON) and non-arteritic anterior ischemic optic neuropathy (NAION), with potentially broad clinical applications in various other neuro-ophthalmic and neurological diseases. Headquartered in Switzerland with operations in the U.S., Iceland and Switzerland, Oculis is led by an experienced management team with a successful track record and supported by leading international healthcare investors.
For more information, please visit: www.oculis.com
Oculis Contact
Ms. Sylvia Cheung, CFO
sylvia.cheung@oculis.com
Investor Relations
LifeSci Advisors
Corey Davis, Ph.D.
cdavis@lifesciadvisors.com
Media Relations
ICR Healthcare
Amber Fennell / David Daley / Sean Leous
oculis@icrhealthcare.com
Cautionary Statement Regarding Forward Looking Statements
This press release contains forward-looking statements and information. For example, statements regarding the potential benefits of the Company’s product candidates, the initiation, timing, progress and results of current and future clinical trials, Oculis’ research and development programs, regulatory and business strategy; Oculis’ future development plans; the timing or likelihood of regulatory filings and approvals; statements about market opportunity, and the Company’s expected financial position and cash runway, are forward-looking. All forward-looking statements are based on estimates and assumptions that, while considered reasonable by Oculis and its management, are inherently uncertain and are inherently subject to risks, variability, and contingencies, many of which are beyond Oculis’ control. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as, and must not be relied on by an investor as, a guarantee, assurance, prediction or definitive statement of a fact or probability. Actual events and circumstances are difficult or impossible to predict and will differ from assumptions. All forward-looking statements are subject to risks, uncertainties and other factors that may cause actual results to differ materially from those that we expected and/or those expressed or implied by such forward-looking statements. Forward-looking statements are subject to numerous conditions, many of which are beyond the control of Oculis, including those set forth in the Risk Factors section of Oculis’ annual report on Form 20-F and any other documents filed with the SEC. Copies of these documents are available on the SEC’s website, www.sec.gov. Oculis undertakes no obligation to update these statements for revisions or changes after the date of this release, except as required by law.
References:
- Ciulla TA, et al. Diabetes Care. 2003;26:2653–2664.
- Petrella RJ, et al. J Ophthalmol. 2012;2012:159167.
- Baker CW, et al. JAMA. 2019;321(19):1880-1894.
- Gonzalez VH, et al. Am J Ophthalmol. 2016;172:72-79.
- Yau et al. Diabetes Care 2012 Mar; 35(3): 556-564
- International Diabetes Federation – diabetesatlas.org
aReal-world data from the American Academy of Ophthalmology (AAO) IRIS Registry. DME, diabetic macular edema; VEGF, vascular endothelial growth factor.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/dc25de9c-07c8-448a-b6af-aea02249e593

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.